Translate this page into:
Antifibrinolytic in reducing postoperative blood loss in total hip replacement and its effect on coagulation profile: A prospective randomized study
Address for correspondence: Dr. Anshul Dahuja, GGS Medical College, Faridkot, Punjab, India. E-mail: anshuldahuja@gmail.com
This article was originally published by Wolters Kluwer - Medknow and was migrated to Scientific Scholar after the change of Publisher.
How to cite this article: Dahuja A, Bhowmik S, Kaur R, Shayam R, Jindal S. Antifibrinolytic in reducing postoperative blood loss in total hip replacement and its effect on coagulation profile: A prospective randomized study. J Orthop Allied Sci 2018;6:3-8.
Abstract
BACKGROUND:
Studies have shown that tranexamic acid reduces blood loss and transfusion need in patients undergoing total hip arthroplasty. However, till date, no study has been large enough to determine definitively whether the drug is safe and effective in total hip arthroplasty. We examined whether intravenous tranexamic acid, when compared with placebo, is safe and effective in total hip arthroplasty.
MATERIALS AND METHODS:
A prospective, randomized, double blinded study was conducted in a group of 142 patients undergoing total hip arthroplasty divided equally into tranexamic acid group and control group. Our protocol included administration of one dose of 15 mg/kg of TXA (given as infusion over 15 min) in 100 ml NS just 15 min before incision and the subsequent 8 hourly in TXA group. In control group we have given 100 ml normal saline infusion just before operation and 100 ml NS infusion 8 hourly for 2 days postoperatively. Postoperative blood parameters were recorded.
RESULTS:
The total postoperative drain output and transfusion requirement was found to be lower in patients who received TXA (352-412 ml) as compared to control group (804-878 ml). We have observed 3 and 4 cases of DVT in TXA and control group respectively. Coagulation profile is least affected in both the groups.
CONCLUSION:
We conclude that tranexamic acid significantly reduces postoperative blood loss and transfusion requirements during total hip arthroplasty.
Keywords
Blood loss
blood transfusion
total hip arthroplasty
tranexamic acid
Introduction
Total hip arthroplasty is associated with extensive postoperative blood loss.[1-7] In the literature studied, mean postoperative blood loss in total hip replacement (THR) was found to vary from 0 to 880 ml and mean total blood loss by 530–1100 ml.[8-10] Increased plasma concentrations of coagulation factors, reduced concentration of coagulation inhibitors, enhanced platelet activity, endovascular release of catecholamines, and serotonin contribute to hemostasis.[8] The fibrinolytic system is activated when tissue trauma releases tissue plasminogen activator (t-PA).[4-6] Thrombin also activates fibrinolysis by triggering release of t-PA from the vascular endothelial cells. Surgical stress enhances the release of plasmin at the site of vascular damage.[6] This may contribute to blood loss after total hip arthroplasty although after surgery, the fibrinolytic system shuts down as a consequence of increased release of plasminogen activator inhibitor that inactivates t-PA[6-10]
Multiple techniques can be used to reduce the risk of perioperative allogenic blood transfusion in THR surgery such as preoperative autologous blood donation, intra- and postoperative red blood cell salvage, controlled hypotension, normovolemic hemodilution, or lowering the transfusion trigger. These techniques have several disadvantages, and they are time consuming, expensive devices are needed, or the poor quality blood quality increases, especially if using postoperatively salvaged but untreated blood.[11] Tranexamic acid (TXA), a synthetic inhibitor of fibrinolysis, competitively blocks a lysine binding site of plasminogen. Plasminogen– TXA complexes redisplaced from the surface of fibrin and as plasmin is prevented from binding to fibrinogen or fibrin monomers, lysis is delayed. At higher concentrations, TXA also acts as a noncompetitive inhibitor of plasmin.[4,5] Although significant reductions in blood loss and transfusion requirements are very consistent in TXA trials for total knee replacement, the results observed in trials for THR show a much greater discrepancy.[12-18] To be effective, the intravenous prophylactic administration of TXA in THR must be made at the beginning of, and not after, the surgical procedure,[17,18] but the wide variability in administration regimens in terms of both dose and duration is a source of concern among orthopedic surgeons.
Despite various studies proving the efficacy of TXA (antifibrinolytic) with single or multiple boluses of different sizes with or without subsequent infusions, no consensus has been reached on the dose of TXA to be administered or duration of treatment.[2,13-15] Claeys et al.[19] postulated that there was no effect on the intraoperative blood loss because TXA delayed lysis of the fibrin clot by the proteolytic action of plasmin, rather than by influencing primary hemostasis and coagulation. He also suggested that a dose of 10 mg/kg would not be sufficient to prevent postoperative bleeding and that higher doses would be required to be effective. However, with a larger dose of 20 mg/kg, the plasma level is maintained for 8 h, which may have the potential to cause prothrombotic complications.[20] The half-life of 1 g of intravenously administered TXA has been found to be 1.9 h, and plasma concentrations remain above the minimum therapeutic level for up to 4 h.[21,22] Three previous studies have investigated the use of a single preoperative dose of 1 g of TXA, and while they have shown a reduction in postoperative blood loss, the sample size was small, the reduction in blood transfusion and cost-benefit were not investigated, and there was no routine investigation for thromboembolism (TE).[14,23,24]
Many studies showed effectiveness of TXA in THA, but no study discussed its effect on coagulation profile. Despite these promising results, valid data on safety are lacking, as large sample sizes are needed to determine this outcome. Thus, concerns about the routine use of TXA remain.
In this study, we have investigated that in patients undergoing total hip arthroplasty, administration of a high dose of TXA for 3 days influences blood loss and whether it has a blood sparing effect along with that we did venography to rule out TE. We also assessed the mechanism of action and effect of TXA on coagulation.
Materials and Methods
From May 2010 to July 2015, 142 adult patients undergoing unilateral primary THR under combined spinal epidural anaesthesia gave informed consent to participate in a double-blind study with a prior randomization. Exclusion criteria were revision THR, a history of venous or arterial TE, thrombophilia, ischemic heart disease, malignancy, a history of epilepsy, and severe chronic renal insufficiency, defined as an estimated glomerular filtration rate >30 mg albumin per gram creatinine in the urine using the modification of diet in renal disease formula. The prospective study was approved by the institutional ethics committee, and written informed consent was obtained from each patient.
A simple randomization schedule using random numbers generated by Microsoft Excel (Microsoft Corp, Redmond, WA) was employed, and after patient eligibility had been confirmed by the anesthetist, the patients were allocated to a group randomized through envelopes opened at anaesthesia induction with 71 patients in TXA group (T group) and 71 patients in control group (C group). The anesthetist was blinded to treatment allocation, as he received the medication from a study nurse, who had prepared the medication before the operation. The operating surgeons were also blinded to the type of TXA treatment. Our protocol included administration of one dose of 15 mg/kg of TXA (given as infusion over 15 min) in 100 ml normal saline (NS) just 15 min before incision and the subsequent 15 mg/kg dose 8 hourly for 48 h postoperatively in tranexamic group. In control group, we have given 100 ml NS infusion just before operation and 100 ml NS infusion 8 hourly for 2 days postoperatively. At the time of postoperative evaluation, neither the patients nor the authors were aware of the group assignments.
Preoperative investigations included hemoglobin (Hb), hematocrit (Hct), a complete coagulogram, liver function tests, renal function test, and routine preanesthetic checkup. Postoperative Hb levels, Hct, and D-dimer were measured 6 h, 24 h after surgery, and at the time of discharge (6–10 days). A negative suction drain was kept for 48 h, and drain output was recorded for day 0 and 1. Blood transfusion in the form of pack cell volume was given to all patients with Hb <9 gm/dl or postoperative drain output >500 ml in first 24 h.
All the operations involved the use of a conventional stemmed unilateral THR performed by a single surgeon through posterior approach. Cemented, uncemented, and hybrid THRs were used depending on bone stock and age of the patient. The method of anaesthesia was either spinal epidural with or without an indwelling catheter or spinal or epidural only, according to the anesthetist's preference. Two negative suction drains (subfascial and subcutaneous) were used in all cases to measure postoperative blood loss and hidden subfascial blood loss.
Postoperative use of deep vein thrombosis (DVT) stockings, ankle pumps, and early mobilization was ensured as a part of thromboprophylaxis. Venous color Doppler was performed at the time of discharge to rule out DVT.
Parameter tests of significance (independent t-test) were used for statistical analysis. Power analysis was also done, and a P < 05 was taken as statistically significant.
Results
The study consists of 142 cases of THR having 71 patients both in control and TXA group. As far as number, age, sex ratio, and diagnosis are concerned, our study is well matched [Flowchart 1].
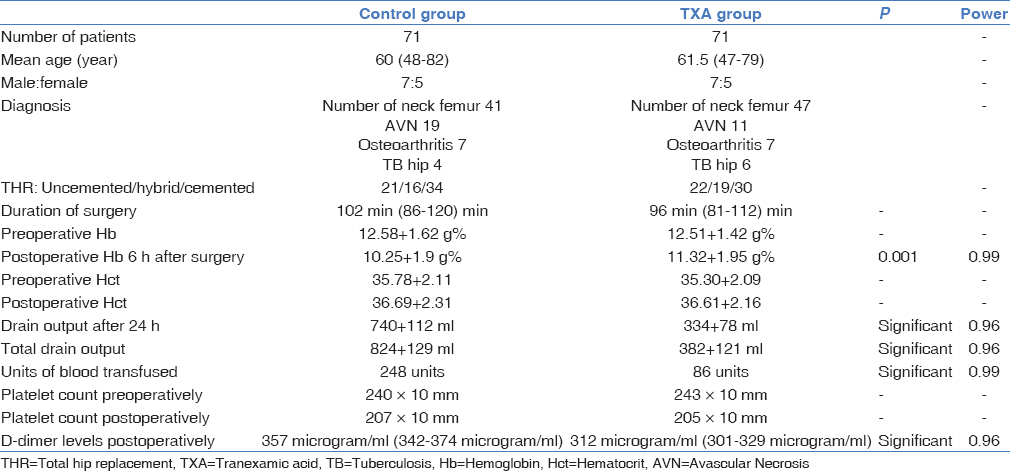
Mean age of the patients undergoing THR was 61.5 years and 60 years in TXA and control group, respectively. Male to female ratio is 7:5 in both the groups. Duration of surgery was 100 min (86–120) for control group as compared to 94 (81–112) min for TXA group.
The mean preoperative Hb and pre- and postoperative Hct values were found to be similar in both the groups. Postoperative Hb was found to be significantly lower in the control group as compared to the TXA group (P = 0.001) and having high power (996).
The total postoperative drain output was found to be lower in patients who received TXA (352–412 ml) as compared to control group (804–878 ml), and this relation was found to be significant with high power concluding a decrease in total blood loss in patients who were administered TXA during total hip arthroplasty.
Above results showed a direct effect on blood transfusion requirement, i.e., tranexamic: control = 1:2.9.
The preoperative and postoperative platelet count were found to be similar in both the groups with a consistent nonsignificant reduction as shown in Table 1.
The preoperative fibrinogen levels were almost similar, but postoperatively after 2 days, they were 623 mg/dl (589–657 mg/dl) in the tranexamic group whereas 577 mg/dl (545–599 mg/dl) in the control group.
The D-dimer levels after 2 days postoperatively were within normal range but found to be higher normal in control group 372 mg/dl (350–380 mg/dl) than tranexamic group 308 mg/dl (302–322 mg/dl).
No adverse effects such as nausea, vomiting, or hypersensitivity were found in any of the patient receiving TXA.
Seven patients (3 in tranexamic group and 4 in control group) developed superficial wound infection for which extended course of antibiotics was given.
Two patients in control group and 1 patient in tranexamic group developed deep infection requiring repeated debridement.
In our study, we found 7 and 5 cases of DVT in control and TXA group. There were no thromboembolic complications in both the groups.
Discussion
The fact that the study was automatically well matched with strong homogeneity of the preoperative data added strength to our study. Various antifibrinolytic agents such as aprotinin, e-aminocaproic acid, and TXA can help reduce blood loss in THA. Of these, TXA is preferred as it is cheaper and less allergenic than aprotinin and is more potent than e-aminocaproic acid.[17] We have evaluated the use of TXA given during total hip arthroplasty and for the first 48 h after operation. TXA decreases blood loss and significantly reduced transfusion requirements. The blood-sparing effect of TXA was most evident during the first 24 h after operation when blood loss was significantly reduced compared with the control group as shown by the drain output. During surgery, the acetabulum is reamed, and the femoral shaft is broached and washed with NaCl under pulsatile pressure. During this period, there is no time for clot formation. This may explain why most authors find no significant reduction in perioperative blood loss.[12,15,16] That is why we just focused on postoperative blood loss. Measurement of postoperative blood loss can also differ depending on the number of drains used postoperatively.
In order to decrease the risk of infection, surgical drains are now removed more rapidly, or a smaller number of drains are placed. The hidden blood loss in hematomas could also add to the discrepancy between the lack of difference in postoperative blood loss and the reduction of patients requiring red blood cell transfusion inTXA-treated patients although studies have shown that the hidden blood loss in THR surgery is small.[19,20]
Fibrinolytic activation is a cascade process that is most easily inhibited in its earlier phase, which may explain why TXA has little effect when given after heavy blood loss.[19,20] TXA inhibits clot lysis more efficiently when administered before clot formation than after the fibrin clot is formed. Once plasminogen is bound to the fibrin surface, TXA is no longer effective.[25] This may explain why TXA has little effect when administered at the end of surgery. Benoni et al. found no significant reduction in postoperative blood loss in total hip arthroplasty when TXA was given toward the end of surgery and that is why we preferred to give TXA just before surgery and then postoperatively.[13]
We found that early blood loss (within 6 h) and total blood loss were significantly reduced in the TXA group. Again, this was consistent with the findings of previous studies and supported the hypothesis that TXA induces inhibition of early fibrinolysis before the body's usual response after 24 h.[14,23,24]
We kept the blood transfusion limit with Hb <9 gm/dl or drain output >500 ml. This study demonstrates that transfusion requirements also reduced in the TXA study group which is comparable with the previous conducted studies.[19,26]
Despite the efficacy of TXA for reducing bleeding, no direct correlation was found between blood loss and variables of fibrinolysis (D-dimer). TXA-treated patients showed no bleeding tendency.
We also found increased concentration of D-dimer in the control group during the first 2 days after operation indirectly support the antifibrinolytic effect of TXA. We did not encounter any increase in thromboembolic activity with TXA. Even a meta-analysis found no increase of thromboembolic complications with the use of TXA in total hip and knee replacement surgery.[26] Benoni et al. suggest that TXA is not associated with an increase in venous thromboembolic events because the effect of TXA is more pronounced in operative wounds than in the peripheral venous blood.[27] He also suggested that TXA does not affect risk of DVT because it inhibits fibrinolysis in the wound not in circulation. Previous research on TXA and thrombosis failed to show any thrombogenic effect,[27] but thrombotic complications were reported with therapy exceeding 24 h.[28] Our data also showed that TXA does not induce platelet activation as platelet count was similar in both the groups postoperatively, and this finding is consistent with the earlier studies.[29]
Limitations of the study include involvement of different anesthetist, different surgeons, different indications for surgery and only postoperative blood loss and postoperative blood transfusion taken into account but still major issues being taken into consideration while performing this study.
Summary
Hence, we conclude that TXA given for 48 h after operation is effective in reducing blood loss and transfusion needs after prosthetic hip surgery. A dose of 15 mg/kg just before operation followed by maintenance dose of 15 mg/kg every 8 h for 24 h would seem appropriate as longer administration of TXA is not accompanied by further reduction in blood loss.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- The prophylactic role of tranexamic acid to reduce blood loss during radical surgery: A prospective study. Anesth Essays Res. 2012;6:70-3.
- [CrossRef] [PubMed] [Google Scholar]
- Tranexamic acid and the reduction of blood loss in total knee and hip arthroplasty: A meta-analysis. BMC Res Notes. 2013;6:184..
- [CrossRef] [PubMed] [Google Scholar]
- Role of tranexamic acid in reducing postoperative blood loss and transfusion requirement in patients undergoing hip and femoral surgeries. Saudi J Anaesth. 2013;7:29-32.
- [CrossRef] [PubMed] [Google Scholar]
- A prospective study on role of tranexamic acid in reducing postoperative blood loss in total knee arthroplasty and its effect on coagulation profile. J Arthroplasty. 2014;29:733-5.
- [CrossRef] [PubMed] [Google Scholar]
- Tranexamic acid use and postoperative outcomes in patients undergoing total hip or knee arthroplasty in the United States: Retrospective analysis of effectiveness and safety. BMJ. 2014;349:g4829.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of extradural and general anaesthesia on the fibrinolytic response to total knee arthroplasty. Br J Anaesth. 1997;79:29-34.
- [CrossRef] [PubMed] [Google Scholar]
- The haemostatic response to surgery and trauma. Br J Anaesth. 1993;70:205-13.
- [CrossRef] [PubMed] [Google Scholar]
- Comparative effects of induced hypotension and normovolaemic haemodilution on blood loss in total hip arthroplasty. Br J Anaesth. 1980;52:1039-43.
- [CrossRef] [PubMed] [Google Scholar]
- Influence of spinal and general anaesthesia on haemostasis during total hip arthroplasty. Br J Anaesth. 1987;59:561-71.
- [CrossRef] [PubMed] [Google Scholar]
- Anesthetic techniques and surgical blood loss in total hip arthroplasty. Acta Anaesthesiol Scand. 1982;26:189-93.
- [CrossRef] [PubMed] [Google Scholar]
- Fibrinolytic activity and postoperative salvaged untreated blood for autologous transfusion in major orthopaedic surgery. Eur J Surg. 2001;167:168-72.
- [CrossRef] [PubMed] [Google Scholar]
- Blood conservation with tranexamic acid in total hip arthroplasty: A randomized, double-blind study in 40 primary operations. Acta Orthop Scand. 2001;72:442-8.
- [CrossRef] [PubMed] [Google Scholar]
- Tranexamic acid, given at the end of the operation, does not reduce postoperative blood loss in hip arthroplasty. Acta Orthop Scand. 2000;71:250-4.
- [CrossRef] [PubMed] [Google Scholar]
- Reduction of blood loss using tranexamic acid in total knee and hip arthroplasties. Arch Orthop Trauma Surg. 2000;120:518-20.
- [CrossRef] [PubMed] [Google Scholar]
- Tranexamic acid reduces blood loss and blood transfusions in primary total hip arthroplasty: A prospective randomized double-blind study in 40 patients. Acta Orthop Scand. 2003;74:665-9.
- [CrossRef] [PubMed] [Google Scholar]
- Tranexamic acid reduces the need for allogenic red blood cell transfusions in patients undergoing total hip replacement. Can J Anaesth. 2004;51:31-7.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical pharmacology of aminocaproic and tranexamic acids. J Clin Pathol Suppl (R Coll Pathol). 1980;14:41-7.
- [CrossRef] [Google Scholar]
- Tranexamic acid reduces blood loss in total hip replacement surgery. Anesth Analg. 2000;91:1124-30.
- [CrossRef] [PubMed] [Google Scholar]
- Reduction of blood loss with tranexamic acid in primary total hip replacement surgery. Acta Chir Belg. 2007;107:397-401.
- [CrossRef] [PubMed] [Google Scholar]
- Application of pharmacokinetic data from healthy volunteers for the prediction of plasma concentrations of tranexamic acid in surgical patients. Clin Drug Invest. 1995;10:280-7.
- [CrossRef] [Google Scholar]
- Single-dose tranexamic acid for reducing bleeding and transfusions in total hip arthroplasty: A double-blind, randomized controlled trial of different doses. Thromb Res. 2016;141:119-23.
- [CrossRef] [PubMed] [Google Scholar]
- One intraoperative dose of tranexamic acid for patients having primary hip or knee arthroplasty. Clin Orthop Relat Res. 2010;468:1905-11.
- [CrossRef] [PubMed] [Google Scholar]
- Tranexamic acid reduces blood loss and financial cost in primary total hip and knee replacement surgery. Orthop Traumatol Surg Res. 2012;98:477-83.
- [CrossRef] [PubMed] [Google Scholar]
- Tranexamic acid reduces blood loss after cementless total hip arthroplasty-prospective randomized study in 40 cases. Int Orthop. 2004;28:69-73.
- [CrossRef] [PubMed] [Google Scholar]
- Application of tranexamic acid in total hip arthroplasty: Current evidences. Ann Trauma Acute Care. 2017;1:1001..
- [Google Scholar]
- Use of intravenous tranexamic acid to reduce allogeneic blood transfusion in total hip and knee arthroplasty: A meta-analysis. Anaesth Intensive Care. 2003;31:529-37.
- [CrossRef] [PubMed] [Google Scholar]
- Fibrinolytic inhibition with tranexamic acid reduces blood loss and blood transfusion after knee arthroplasty: A prospective, randomised, double-blind study of 86 patients. J Bone Joint Surg Br. 1996;78:434-40.
- [CrossRef] [Google Scholar]
- Massive pulmonary thromboembolism after tranexamic acid antifibrinolytic therapy. Br J Clin Pract. 1989;43:465-6.
- [CrossRef] [Google Scholar]
- Reversible translocation of glycoprotein Ib in plasmin-treated platelets: Consequences for platelet function. Eur J Clin Invest. 1993;23:785-93.
- [CrossRef] [PubMed] [Google Scholar]






